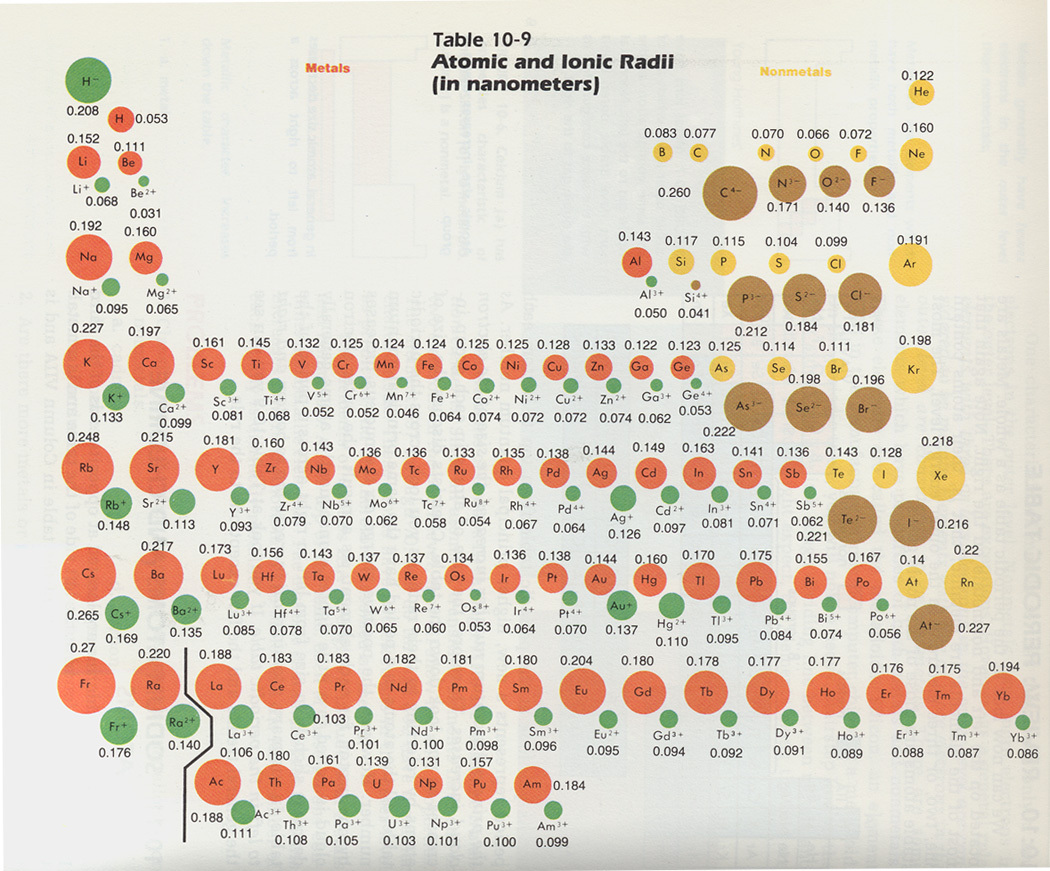
Ductility, thermal conductivity, metallic lustre, malleability and high electrical and thermal conductivity are some of the properties of metals. This implies that they have a metallic character. Most of the elements in this table are metals. Noble gas elements have almost zero electron affinity. The elements present within a group contain similar affinity. This is mainly dependent on the arrangement of the outer electron shell. It is also the energy released on the removal of a single-charged anion. The value of an electron affinity in an atom is basically the energy acquired on the addition of an electron. It declines moving down a column and rises while moving from left to right in a periodic table. Here, the former is closer to the nucleus and thus the size of the atom declines.Įlectron Affinity means how swiftly an atom accepts an electron. As one goes across a row of the periodic table, a greater number of electrons and protons can be observed. These elements gain an electron energy shell and hence the atoms become larger. Ionic radius, on the other hand, is the distance amid two atomic nuclei touching one another.Īs one goes downwards in the periodic table, elements with more protons are seen. This is basically the distance covered from the outermost stable electron to the nucleus.

The electronegativity of atoms is affected by the distance between the nucleus and the valence electrons and the atomic number. Two atoms have a greater chance of forming a chemical bond when the difference between their electronegativity values is larger. However, there are exceptions to it at times. It sees a decreasing trend when you move down a group. Electronegativity usually rises from left to right. It shows how an atom can swiftly form a chemical bond. Periodic Table TrendsĮlectronegativity is the ability of an atom to pull electrons towards it. Here is a look at the periodic table trends of electronegativity, atomic radius, electron affinity, metallic character and ionization energy. This makes the recurring element properties noticeable in this table. In the periodic table, elements are categorized based on their electronic structure.
